ISO 15223-1: 5.1. Manufacture
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Manufacturer | 5.1.1. | Indicates the medical device manufacturer. |
 | Authorized representative in the European Union | 5.1.2. | Indicates the authorized representative in the European Union. |
 | Date of manufacture | 5.1.3. | Indicates the date when the medical device was manufactured. |
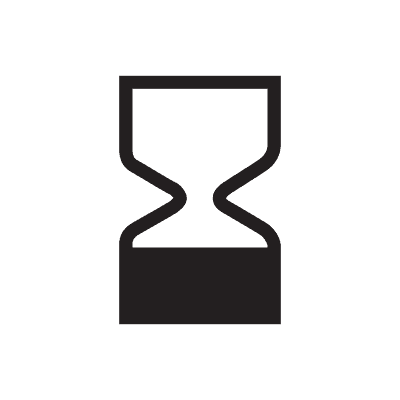 | Use-by date | 5.1.4. | Indicates the date after which the medical device is not to be used. NOTE: Synonyms for “use-by date” are “use by”, “expiry date” and “expiration date”. |
 | Lot number | 5.1.5. | Indicates the manufacturer’s batch code so that the batch or lot can be identified. |
 | Catalog number | 5.1.6. | Indicates the manufacturer’s catalogue number so that the medical device can be identified. NOTE: Synonyms for “catalog number” are “commercial product name”, “commercial product code”, stock keeping unit, “reference number” and “reorder number”. |
 | Importer | 5.1.8. | Indicates the entity importing the medical device into the locale. |
 | Serial number | 5.1.7. | Indicates the manufacturer’s serial number so that a specific medical device can be identified. |
ISO 15223-1: 5.2. Sterility
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Sterilized using ethylene oxide | 5.2.3. | Indicates a medical device that has been sterilized using ethylene oxide. |
 | Do Not Resterilize | 5.2.6. | Indicates a medical device that is not to be resterilized. |
 | Non-Sterile | 5.2.7. | Indicates a medical device that has not been subjected to a sterilization process. |
 | Do not use if package is damaged and consult instructions for use | 5.2.8. | Indicates a medical device that should not be used if the package has been damaged or opened. NOTE 1: This symbol can also mean “Do not use if the product sterile barrier system or its packaging is compromised”. |
 | Single sterile barrier system | 5.2.11. | Indicates a single sterile barrier system. |
ISO 15223-1: 5.3. Storage
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Fragile, handle with care | 5.3.1. | Indicates a medical device that can be broken or damaged if not handled carefully. |
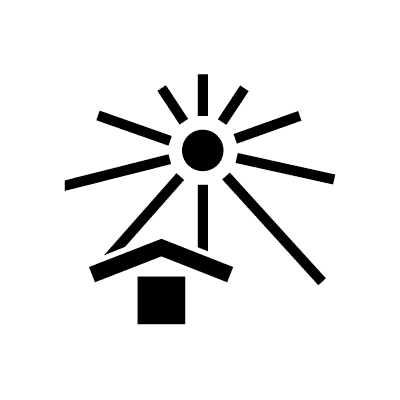 | Keep away from sunlight | 5.3.2. | Indicates a medical device that needs protection from light sources. NOTE: This symbol can also mean “Keep away from heat”. |
 | Keep dry | 5.3.4. | Indicates a medical device that needs to be protected from moisture. |
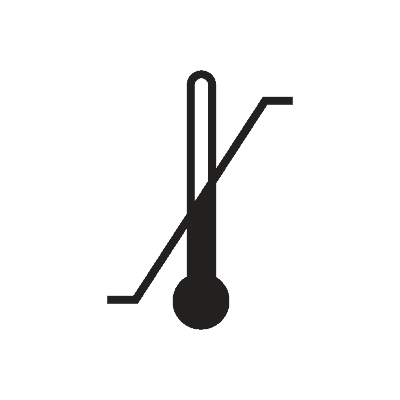 | Temperature limit | 5.3.7. | Indicates the temperature limits to which the medical device can be safely exposed. |
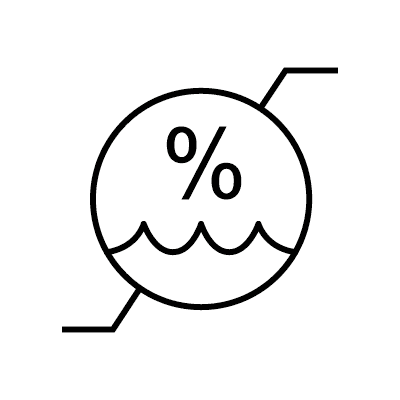 | Humidity limitation | 5.3.8. | Indicates the range of humidity to which the medical device can be safely exposed. |
ISO 15223-1: 5.4. Safe use
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Do not re-use | 5.4.2. | Indicates a medical device that is intended for one single use only. |
 | Consult instructions for use | 5.4.3. A.16 NOTE | A.16 Example of use of symbol 5.4.3, “Consult instructions for use” for an electronic instruction for use (eIFU) NOTE: The eIFU indicator can be a manufacturer’s website URL or some other appropriate indication that the instructions for use are available in an electronic format. |
 | Caution | 5.4.4. | To indicate that caution is necessary when operating the device or control close to where the symbol is placed, or to indicate that the current situation needs operator awareness or operator action in order to avoid undesirable consequences. |
 | Not made with bis (2-ethylhexyl) phthalate (DEHP) | BS EN 15986:2011 EN 15986:2011(E) Annex B | |
 | Contains or presence of phthalate": bis (2-ethylhexyl) phthalate (DEHP) | BS EN 15986:2011 EN 15986:2011(E) Clause 4.2 Annex A | |
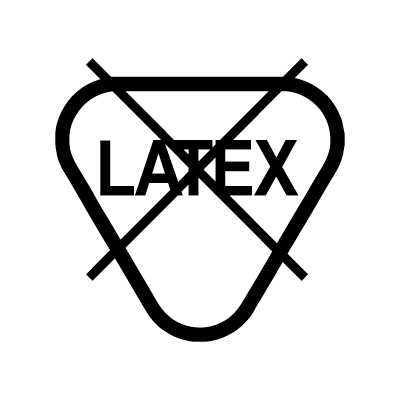 | Not made with natural rubber latex | ||
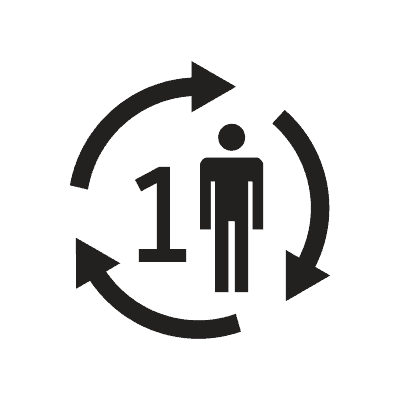 | Single patient-multiple use | 5.4.12. | Indicates a medical device that may be used multiple times (multiple procedures) on a single patient. |
ISO 15223-1: 5.6. Transfusion/infusion
| Symbol | Title | Reference | Description |
|---|---|---|---|
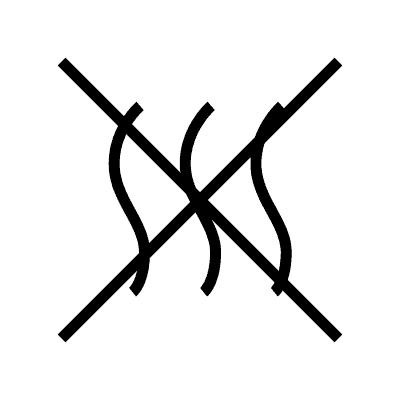 | Non-pyrogenic | 5.6.3. | Indicates a medical device that is non-pyrogenic. |
ISO 15223-1: 5.7. Others
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Medical device | 5.7.7. | Indicates the item is a medical device (under EU Legislation). |
 | Unique Device Identifier | 5.7.10. | Indicates a carrier that contains Unique Device Identifier information. |
IEC 60601-1: General Symbols
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Type BF applied part | Table D.1, Symbol 20 | To identify a type BF applied part complying with IEC 60601-1. |
 | Follow instructions for use | Table D.2, Symbol 10 | Symbol must appear in color. Limited to Medical Electrical Equipment covered by EN 60601-1. |
IEC 60601-1: Degrees of Protection
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | Finger-Sized Object & Tilted Drip Water Protection | Table D.3, Symbol 2 | Indicates that product enclosure is protected against solid foreign objects of 12,5 mm and greater; and has protection against vertically falling water drops when ENCLOSURE tilted up to 15°. |
ASTM F 2503: Magnetic Resonance
| Symbol | Title | Reference | Description |
|---|---|---|---|
 | MR Unsafe | ASTM F2503-20: 7.3.3 | An item which poses unacceptable risks to the patient, medical staff or other persons within the MR environment. (ASTM F2503-20:3.1.14) |
Other Symbols
| Symbol | Title | Reference | Description |
|---|---|---|---|
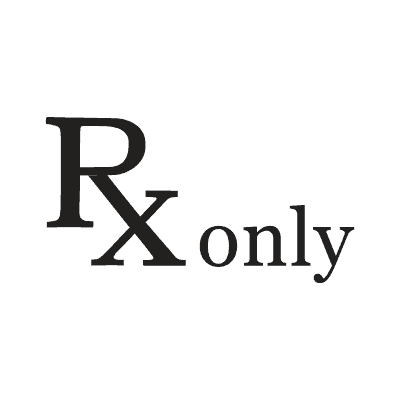 | Prescription Only | 21 CFR 801.109 (FDA) | Caution: Federal law restricts this device to sale by or on the order of a physician |
 | CE Mark | Conformity marking indicating that a product meets European Union (EU) safety, health, and environmental protection requirements. |

